2024 Highlights
Montefiore Einstein Neurology
World-renowned Neurologists.
Elite Biomedical Scientists.
Life-changing Results.
The Saul R. Korey Department of Neurology created a visionary model of neurological care that enabled us to envision neurological diseases as lifespan disorders, forever changing the lens through which neurological diseases are understood.
Childhood and early onset disorders necessitate different treatment modalities as the brain changes during adult life and aging. As medical breakthroughs have increased the median survival, our institution offers a destination site reflecting the aspiration that these patients can lead increasingly productive and fulfilling lives as they age. Research carried out in departmental laboratories has led to the paradigm-shifting discovery that aging likely starts at conception, and that the forerunners of late-onset dementias had their origin at the early stages of life, and account for the fact that these disorders in fact are systemic disorders that need to be treated in an interdisciplinary way. These revolutionary discoveries have profound implications for the early treatment of these currently untreatable conditions using a range of stem cell, epigenetic reprogramming and dynamic tissue remodeling strategies, thereby offering hope for the development of true disease-modifying treatments and even disease cures.
DATA & STATISTICS
2024 by the numbers
27,882
outpatient
31,562
inpatient
6,196
admissions
12,133
procedures
HIGHLIGHTS
Reimagining the Frontiers of Neurology Innovations by Igniting a Revolution in Understanding Disease Causality and Therapeutic Breakthroughs
A collaborative effort between the NIH and Montefiore Einstein, the Icahn School of Medicine at Mount Sinai, New York University School of Medicine and other affiliated institutions and partner sites, our Regional Coordinating Center encompasses all boroughs of New York City, Westchester, Long Island and Upstate New York, as well as Nashville, TN, and we continue to expand our network, leadership, and influence.
CLINICAL TRIALS
Transforming the landscape of clinical care
At the Saul R. Korey Department of Neurology, we are committed to finding new and innovative options to prevent, diagnose, treat and cure neurologic diseases.
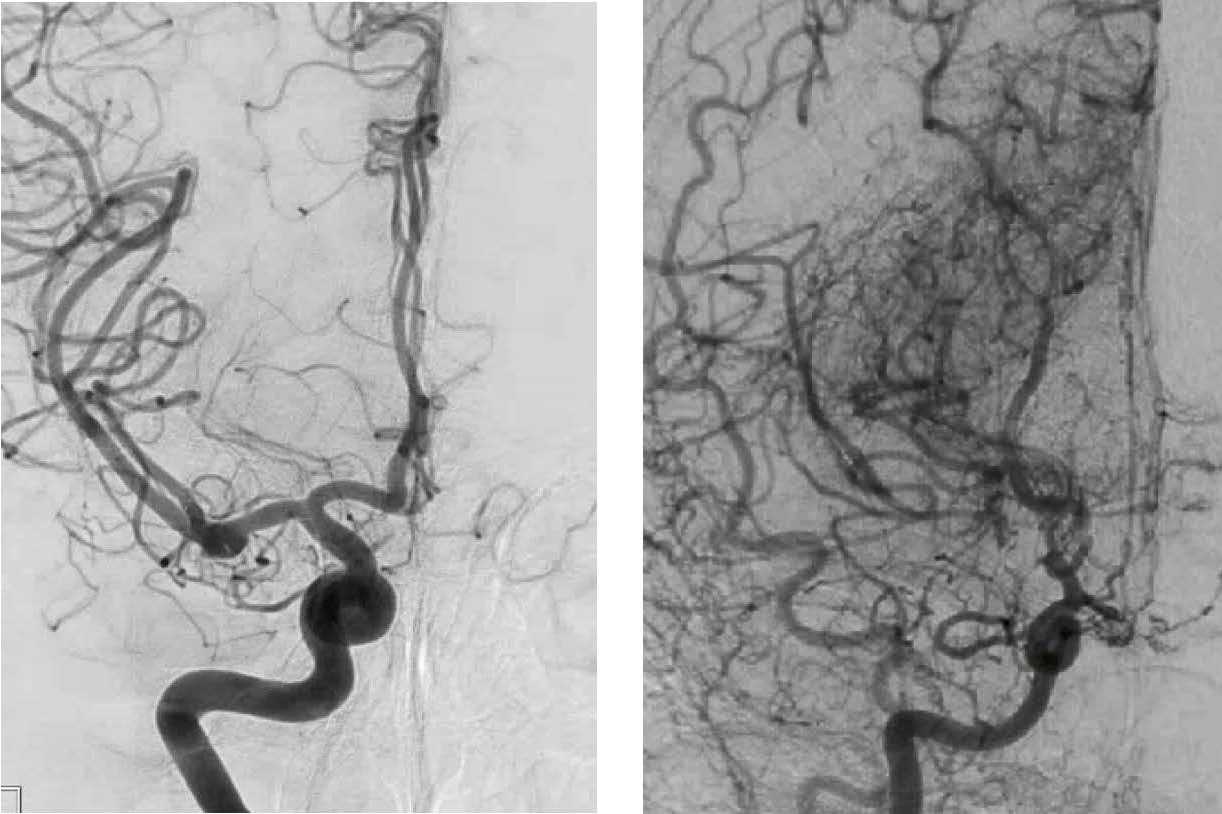
We are spearheading numerous clinical trials to advance treatments and receive notable NIH-funded grants, including our elite NINDS/NIH StrokeNet neurovascular clinical trials consortium grant.
We use these grants for the study of a wide range of neurological diseases including dementia, Alzheimer’s disease, epilepsy, Multiple Sclerosis, and Huntington’s disease, as well as research that examines novel ways to restore brain function and studies that target the effects of COVID-19 on the brain in those with mild or asymptomatic infection.
International Workshop: What is Needed to Ensure Outcome Measures for Rett Syndrome are Fit-for-purpose for Clinical Trials?
SleepSMART: Sleep for Stroke Management and Recovery Trial
ASPIRE: Anticoagulation for Stroke Prevention and Recovery after ICH
SATURN: Statins Use in Intracerebral Hemorrhage Patient
CAPTIVA: Comparison of Anti-coagulation and Anti-platelet Therapies for Intracranial Vascular Atherostenosis
DISCOVERY: Determinants of Incident Stroke Cognitive Outcomes and Vascular Effects on RecoverY
VERIFY: Validation of Early Prognostic Data for Recovery Outcome after Stroke for Future, Higher Yield Trials
The ALLIANCE Aortic Valve-in-valve (AViV) Trial will Evaluate if the SAPIEN X4 Transcatheter Heart Valve (THV) is Beneficial for Patients with a Failing Aortic Bioprosthetic Valve
A Phase 2/3 Multicenter, Randomized, Double-blind, Placebo-controlled Study to Evaluate the Efficacy, Safety, and Tolerability of BHV-7000
Phase 3, Randomized, Double-Blind, Placebo-Controlled Study with an Open-label Extension Period to Evaluate the Efficacy and Safety of Telitacicept
Team News
Updates and additions to our world-renowned faculty
Patient referrals
At Montefiore Einstein Saul R. Korey Department of Neurology, we know providing patients with the best possible care includes teamwork and trust. We work closely with our valued referring physicians to ensure open communication and reliable expertise.
Contact Us
Mark F. Mehler MD, Chair, Neurology
mark.mehler@einsteinmed.edu